AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
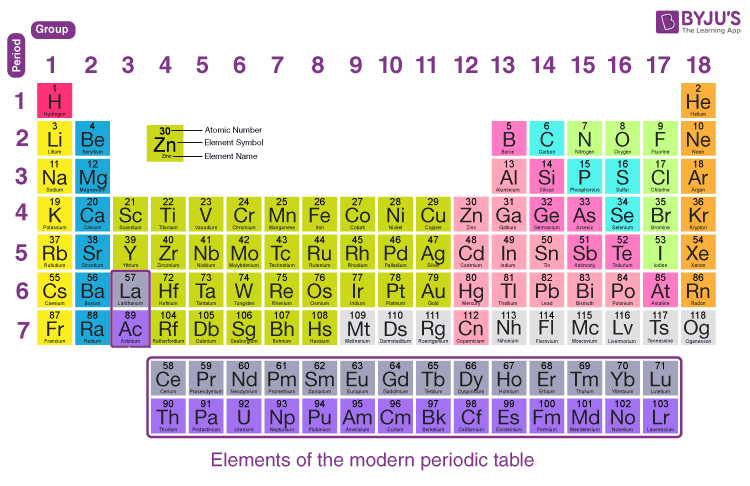
 We just place it there because it is unique and does not really fit into any group. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Helium is not truly a member of Group 18 (or Group VIII in the older system). Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Free Gift for you: Interactive Periodic Table PVC (polyvinyl chloride) is the highly used polymer which contains chlorine in it.Chlorine is added to the swimming pool water to keep it clean.It is also used in manufacturing of medicines, disinfectants as well as insecticides.Chlorine element is used in manufacturing of paper, plastics, textiles, dies, etc.In earlier days, chlorine was used as a chemical weapon as it can burn the skin.The chlorine present in CFC destroys the ozone molecules and hence it should be handled very carefully.Electrolysis of salt water can produce chlorine gas.Chlorine is also used to make drinking water safe from microorganisms.Hence the leakage of chlorine gas from the containers can be detected by using ammonia in this way. Chlorine reacts with ammonia and forms white mist.Chlorine is a non explosive and non flammable gas.Chlorine has high electronegativity and higher electron affinity, so it reacts with almost all the elements except with lighter noble gases.Chlorine is chemically reactive and hence it is not found in a free state, but it is always found as a compound with other elements (for example, NaCl, HCl, etc).Chemical properties of ChlorineĬhemical properties of chlorine are mentioned below. Density of chlorine gas is 3.214 g/L which is far more than the density of the atmospheric gases of the earth.The melting point of diatomic chlorine gas (Cl 2) is -101.5 ☌ and it’s boiling point is -34.04 ☌.Chlorine is a pale yellow-green colored gas having a strong smell.Physical properties of chlorine are mentioned below. 
The physical and chemical properties of chlorine element are mentioned below.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed